Country Selector
Please enter a valid location
- Key points
- Micronutrients are essential
- Plant requirements and soil availability
- Organic matter
- Micronutrient removal by crops
- Detecting micronutrient deficiencies
- Managing micronutrient deficiencies
- References
Key points
- Micronutrients are seven elements essential for crop growth in very low quantities – boron, chloride, copper, iron, manganese, molybdenum, and zinc.
- In the major crops and production areas of North America, the micronutrients most often supplied by fertilization include zinc, manganese, boron, and iron.
- Micronutrient deficiencies can be detected by visual symptoms on crops and by testing soils and plant tissues.
- The most reliable micronutrient soil tests are for zinc, boron, copper, and manganese. Though useful, these tests are not as precise as those for soil pH, potassium and phosphorus.
- Plant tissue analysis is more reliable than soil testing for identifying many micronutrient problems and can also supplement soil test information.
- Most often, micronutrients are soil-applied in a band at planting, or foliar-applied, as these methods allow lower use rates of sometimes expensive materials.
Micronutrients are essential
Micronutrients are essential elements that are used by plants in small quantities. For most micronutrients, crop uptake is less than one pound per acre. Despite this low requirement, critical plant functions can be limited if micronutrients are deficient, resulting in plant abnormalities, reduced growth and lower yield. In such cases, expensive, high requirement crop inputs such as nitrogen and water may be wasted if yield potential is being limited by a micronutrient deficiency. This Crop Insights will discuss general micronutrient requirements, deficiency symptoms, soil and plant sampling, and fertilization practices.
Plant requirements and soil availability
There are 16 elements essential to growth of crop plants (Figure 1). Two of these nutrients, carbon and oxygen, are extracted from the air. Hydrogen is extracted from soil water. The remaining thirteen nutrients are all extracted from soil and are classified as primary macronutrients, secondary macronutrients, or micronutrients based on the quantities taken up and utilized for plant growth. The seven micronutrients – boron, chloride, copper, iron, manganese, molybdenum, and zinc – are used in very low quantities for crop production (Figure 2).
Image 1. - Corn leaves showing zinc deficiency. Interveinal striping in center of leaf is surrounded by green borders/margins.

Figure 1. Sources of the sixteen nutrients essential for crop production.
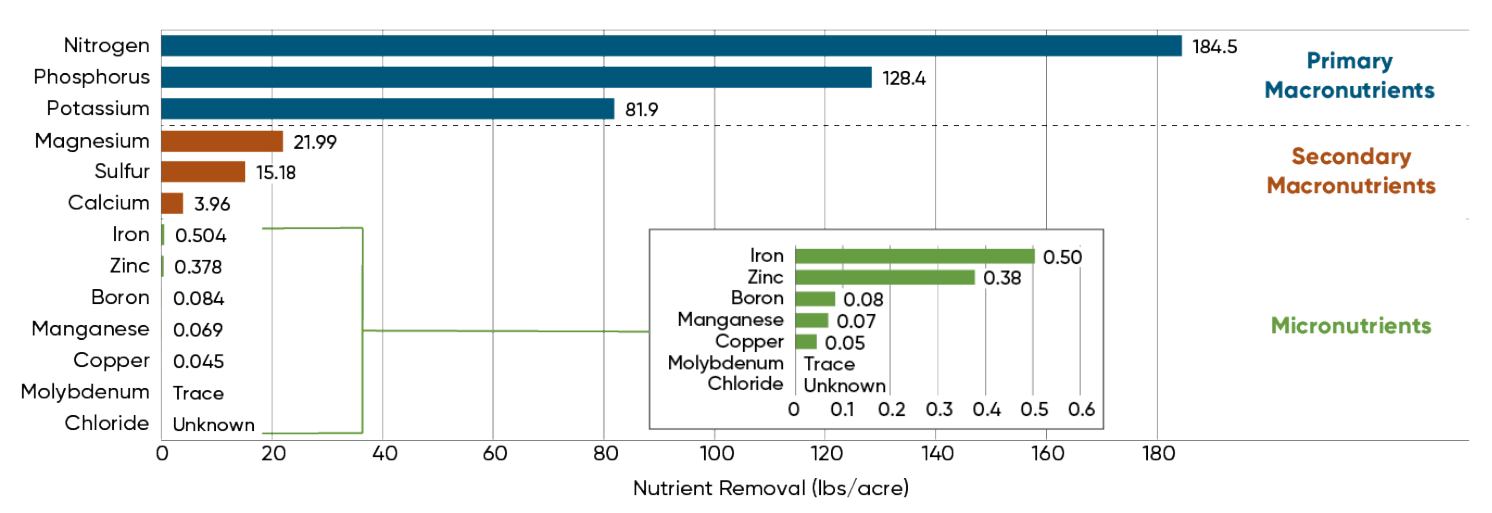
Figure 2. Nutrients removed by a 300 bu/acre crop (Heckman et al., 2003).
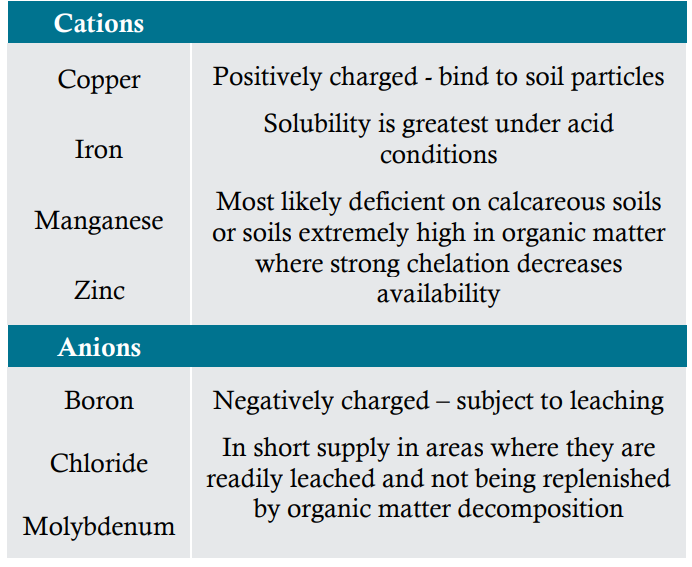
The seven micronutrients are sufficient in most soils to meet crop needs. However, some sandy soils and other low-organic matter soils are naturally low in micronutrients, and high pH soils may make some micronutrients less available and therefore deficient. In the major crops and production areas of North America, the micronutrients most often supplied by fertilization include zinc, manganese, boron, and iron. Basic chemical properties of micronutrients help determine their availability in soils (Table 1).
Table 1 – Chemical properties of micronutrients
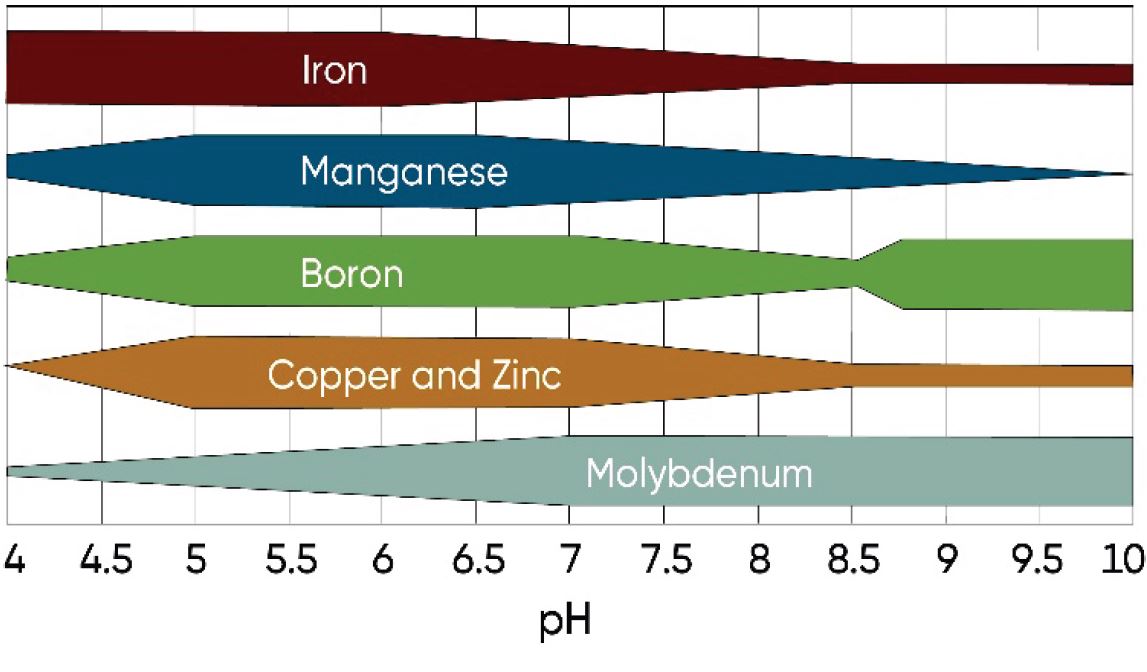
Because of complex chemical reactions within the soil, micronutrient availability is ultimately controlled by the equilibrium between the soil solution, soil organic matter, cation exchange sites, and insoluble compounds of micro-nutrients. Soil acidity or alkalinity has a large effect on the tie-up of micronutrients or their availability to plants. Micronutrients are generally more available in acid soils and less available at high pH, with the exception of molybdenum, which is more available at higher pH (Figure 3).
Organic matter
Organic matter is a reservoir for essential plant nutrients, continuously supplying these nutrients to the crop as it decomposes over time. This reservoir is especially important for anions such as boron, which do not bind to soil particles and are therefore subject to losses. Soils that receive regular additions of organic residues such as manures rarely show micronutrient deficiencies. An exception is deficiencies caused by nutrient imbalances, such as a deficiency of manganese caused by an excess of phosphorus in overly manured soils. Another exception is soils with extremely high organic matter such as muck or peat soils. In these soils, strong, natural chelation (the combination of a micronutrient with an organic molecule) can make some micronutrients unavailable, particularly copper, manganese, and zinc.
Figure 3. Relative availability of micronutrients by soil pH1
Micronutrient removal by crops
Crop yields are continually increasing due to genetic improvements in stress tolerance and disease resistance, incorporation of insect resistance traits, and use of seed treatments and other crop protection products. This means that more micronutrients are removed from the soil as yields increase. Estimates of nutrient removal for a 300 bu/acre corn crop are shown in Figure 2.
Although micronutrient removal rates are increasing, they are still very small relative to the primary and secondary macronutrients. Removal rates for a 300 bu/acre corn crop range from over 80 lbs/acre for the primary macronutrients and 4-22 lbs/acre for secondary macronutrients, compared to 0.5 lb/acre or less for micronutrients.

Image 2 - Symptoms of boron deficiency in alfalfa. Alfalfa is one of the few crops that can benefit from boron applications if levels become deficient.
Detecting micronutrient deficiencies
Micronutrient deficiencies can be detected by visual symptoms on crops and by testing soils and plant tissues. To understand visual symptoms, it is useful to know the role each micronutrient plays in plant growth and development.
Functions of Micronutrients
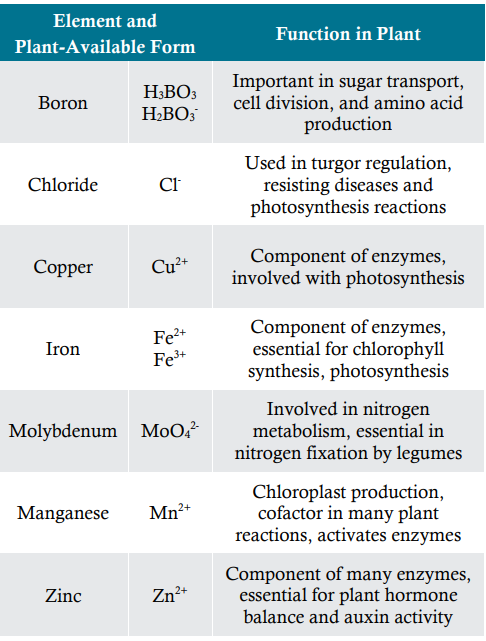
Micronutrients differ in the form they are absorbed by the plant, their functions and mobility in the plant, and their characteristic deficiency or toxicity symptoms (Table 2 and 3).
Table 2. Plant available forms and functions of micro-nutrients in plants2.
Micronutrient Deficiency Symptoms
Except for Mo, the micronutrients are considered weakly mobile or immobile in plants. This means that deficiency symptoms appear first or most severely on newest plant tissues. For molybdenum, deficiency symptoms appear first on oldest plant tissues. Symptoms vary according to crop, but generalized symptoms are shown in Table 3.
Table 3. General micronutrient deficiency symptoms2.

Image 3 - Iron deficiency chlorosis (IDC) of soybeans caused by high pH soils in the Black Belt region of central Alabama. IDC is a complex plant disorder associated with high pH soils and soils containing soluble salts where chemical conditions reduce the availability of iron.
Micronutrient deficiencies usually have a patchy distribution in fields due to variation in soil properties that affect availability (e.g., pH, drainage, and salinity) and management history such as manure applications. Learning to visually identify deficiencies is important in recognizing problem areas and planning remediation for future crops. However, it is often too late for corrective action in the current crop by the time visual symptoms appear.
Common Micronutrient Deficiencies
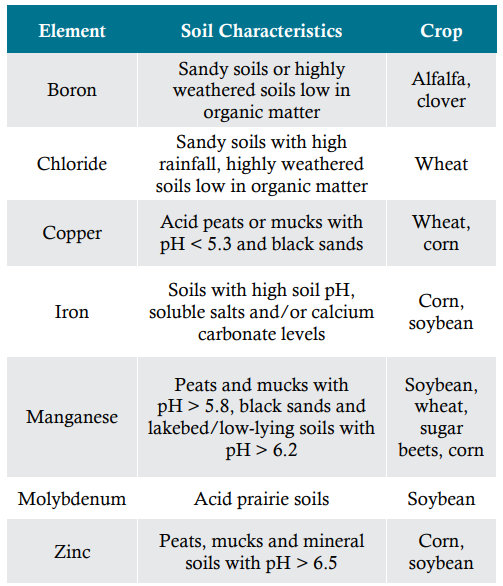
Micronutrient deficiencies tend to appear with much greater frequency on specific soil types and in certain crops (Table 4).
Table 4. Soil conditions which may lead to micronutrient deficiencies for various crops3.
Soil Tests to Detect Micronutrient Deficiencies
Many plant symptoms associated with micronutrient deficiencies, including stunting and chlorosis, may have a variety of causes, including disease, insect or herbicide damage, or environmental conditions. Soil and plant analysis are both useful in determining if the cause is truly nutritional. Though adequate for this purpose, micronutrient soil tests are not as precise as soil pH, phosphorus, and potassium tests.
The most reliable micronutrient soil tests are for zinc, boron, copper, and manganese. Because interpretations are soil specific, it is best to use locally calibrated recommendations. Soil tests for iron and molybdenum are considered to be of little value in predicting the supply of these nutrients in soils. When sampling for micronutrients, sample the root zone down to 8 inches deep.
Plant Analysis to Detect Micronutrient Deficiencies
Plant tissue analysis is more reliable than soil testing for identifying many micronutrient problems and can also supplement soil test information. Tissue testing is especially valuable in cases where reliable soil tests are unavailable. However, molybdenum and chloride levels cannot be determined by this method.
Plant analysis can be used in two ways; one is to monitor the crop's micronutrient status, and the other is to diagnose a problem situation. By quantifying the nutrient content of tissues, plant analysis can point out an existing or potential problem before visual symptoms develop.
If in-season micronutrient deficiencies are suspected, plant samples should be taken as early as practical; treatments, when needed, should be made in a timely manner. Research has shown that once a micronutrient deficiency is detected, the plant has already suffered irreversible yield loss.
Because plant nutrient composition varies with the crop, age of the plant, part of the plant sampled and other factors, it is important to follow the standard sampling procedures provided by your plant diagnostic laboratory. In order to obtain a representative sample, take multiple plants from areas randomly distributed throughout the affected field area. Avoid border plants and those contaminated with dust, soil or foliar sprays. Taking samples of non-symptomatic plants to compare with apparent nutrient-deficient plants can increase the usefulness of plant analysis. Be aware that interpreting results is complex and may require expert advice.
Managing micronutrient deficiencies
Selecting Micronutrient Sources
There are three main classes of micronutrient fertilizers: inorganic, synthetic chelates, and natural organic complexes.
Inorganic sources consist of oxides, carbonates, and metallic salts such as sulfates, chlorides, and nitrates. Sulfates are the most common metallic salts used in the fertilizer industry because of their high water-solubility and plant availability. Less soluble oxides must be finely ground or partially acidulated with sulfuric acid to form oxysulfates in order to increase their effectiveness. Metal-ammonia complexes such as ammoniated Zn sulfate decompose readily in soils and provide good agronomic effectiveness.
Chelates are fertilizers in which the micronutrient is combined with an organic molecule to increase its stability and effectiveness in the soil. Chelates such as Zn-EDTA are more stable and more effective in correcting Zn deficiency than other forms of applied Zn. Synthetic chelates are more effective and less variable than natural organic complexes such as lignosulfates, phenols, and polyflavonoids.
Method of Application
The best method of micronutrient application depends on the element and when the deficiency is being addressed.
Soil application. For deficiencies known at the start of the season, soil application is preferred to foliar application for most nutrients. Micronutrients banded with starter fertilizers at planting time are usually more effective over a longer period than foliar-applied micronutrients. This method also gets the nutrient to the plant at the earliest opportunity.
Soil-applied micronutrients may also be broadcast, but a concentrated band near the plant allows lower use rates of sometimes expensive materials. Manganese should only be banded, because of the ability of most soils to strongly “fix” this element. However, boron should not be banded, as high concentrations near the seed can be toxic.
Foliar application is especially useful for some elements that are not efficiently used when applied to the soil, such as iron. This method is also useful for quick uptake in emergency situations when deficiencies are noted or in cases where other materials are being sprayed. Like banding, foliar applications generally have lower use rates, but more than one application may be needed. However, because the crop partially develops prior to foliar application, irreversible damage may have already occurred before the needed nutrient is supplied.
Broad-spectrum micronutrient applications are not recommended to treat a single micronutrient deficiency, as this approach is expensive and potentially harmful to the crop. The harm can occur because of potential toxicities, or because the presence of additional nutrients may interfere with the uptake of the needed nutrient.
Achieving a uniform spread pattern is important to correct deficiencies, regardless of whether the material is liquid or solid, banded or broadcast, or preplant or foliar applied.
References
1 Adapted from E. Truog. 1946. Soil reaction influence on availability of plant nutrients. Soil Science Society of America Proceedings 11, 305-308.
2 Adapted from W.F. Bennett (editor), 1993. Nutrient deficiencies and toxicities in Crop Plants, APS Press, St. Paul, MN.
3 Adapted from Tri-State Fertilizer Recommendations for Corn, Soybeans, Wheat and Alfalfa, Ohio State University. Online at: https://agcrops.osu.edu/FertilityResources/tri-state_info
Heckman, J.R., J.T. Sims, D.B. Beegle, F.J. Coale, S.J. Herbert, T.W. Bruulsema, and W.J. Bamka. 2003. Nutrient removal by corn grain harvest. Agronomy J. 95:587-591.
Kelling, K. 2005. Micronutrient management in the north-central US and Canada. University of Wisconsin, Madison. Presented at the llth Annual Southwest Agricultural Conference, Ridgetown, ONT.
Stevens, G., P. Motavalli, P. Scharf, M. Nathan, and D. Dunn. 2018. Crop nutrient deficiencies and toxicities. University of Missouri-Columbia Extension pub. IPM 1016. Online at: https://extension.missouri.edu/ media/wysiwyg/Extensiondata/Pub/pdf/agguides/pests/ipm1016.pdf