Country Selector
Please enter a valid location
Plant Physiology in Response to Pathogen Attack
Crop Insights from Pioneer Agronomy Sciences
- Summary
- Introduction
- Types of Pathogens
- Types of Defense
- Requirements for Infection
- Plant Detection and Response to Pathogens
- Structural Defenses
- Chemical Defense
- Proteins and Enzymes
- Qualitative vs Quantitative Resistance
- Pathogen Resistance
- Conclusions
- References
Summary
- Genetic resistance of crop cultivars to plant diseases is an important tool to protect crop yield and quality.
- Plant pathogens can differ in their infection tactics – they can feed off living plant tissue, kill tissue of the host plant and feed of the components of the dead tissue, or both.
- Plants can utilize several strategies to defend themselves against pathogens, including defenses that are always present and defenses that can be activated in response to pathogens.
- Resistance can be expressed genetically as traits that are either qualitative (involving one mechanism) or quantitative (involving many mechanisms).
- The challenge for improvement of crop varieties is determining how quantitative resistance genes apply to a specific pathogen.
Introduction
Plant pathogens are one of the most important threats to crop production globally. Potential yield losses from pathogens are estimated at around 16 percent on average (Oerke, 2005); however, severe infections can wipe out, contaminate, or decrease the quality of agricultural products. Genetic resistance to diseases in crop cultivars has played a key role in the battle against fungi, bacteria, and viruses; providing an important line of defense for protecting crop yield and quality from crop diseases. Plant diseases vary in the mechanisms that they use to infect plants and plant species have evolved a wide range of defenses to protect them from pathogens. Plant breeders work to incorporate these natural defense mechanisms into crop cultivars, making them more resistant to common crop diseases.
This Crop Insights reviews some of the physiology behind plant resistance to pathogens – how pathogens attack plants, how plants defend themselves, and how pathogens are able to sometimes overcome these plant defenses. The ongoing evolutionary arms race between plants and pathogens demonstrates the complexity of evolutionary biology, and how these molecular battles can directly impact our crop production and food supply.

Corn field severely infected by northern corn leaf blight.
Types of Pathogens
Pathogens are divided into three general categories according to mechanisms they employ to colonize and feed off of plants:
Biotrophs feed on living host tissue. Biotrophic pathogens establish specialized biochemical connections with the host plant during infection. These communications suppress the plants defense and increase the quantity of nutrients being sent to the infection site. Examples of biotrophic pathogens in corn include common rust (Puccinia sorghi), southern rust (Puccinia polysora), tar spot (Phyllachora maydis), and Physoderma brown spot (Physoderma maydis).
Necrotrophs kill host tissue and feed off of the dead tissue. They accomplish this by quickly overwhelming the plant with toxic compounds and tissue-degrading enzymes before the host plant is able to react and initiate defenses. Examples of necrotrophic pathogens in corn include gray leaf spot (Cercospora zeae-maydis), and southern leaf blight (Cochliobolus heterostrophus).
Hemibiotrophs utilize components of both strategies. They begin as biotrophic during initial infection and become necrotrophic as colonization progresses. Examples of hemibiotrophic pathogens in corn include northern corn leaf blight (Exserohilum turcicum) and anthracnose (Colletotrichum graminicola).

Southern rust establishes a biotrophic relationship with corn (left), whereas gray leaf spot uses a necrotrophic approach (right).
Types of Defense
Plant species have evolved a wide range of different mechanisms to protect themselves from invading pathogens that can be classified into two general categories:
Constitutive defense is a form of continuous defense that the plant is able to utilize without expending additional energy. Constitutive defense includes barriers like cell walls, waxy cuticles, and bark that deter pathogens from invading the plant.
Inducible defenses are turned on after the pathogen is detected by the plant. Plants can produce toxic chemicals, deploy pathogen-degrading enzymes, or destroy infected plant cells. Inducible defenses remain latent because they require energy and nutrients to activate and maintain.
Requirements for Infection
For disease to occur, three things must be present: a susceptible host plant, a pathogen, and an environment favorable for infection. For many common crop diseases that are able to overwinter in crop residue, the pathogen is always present at some level in fields, making environmental conditions during the growing season the key determinant for infection. Diseases vary in the specific conditions that are optimal for the pathogens to commence infection.
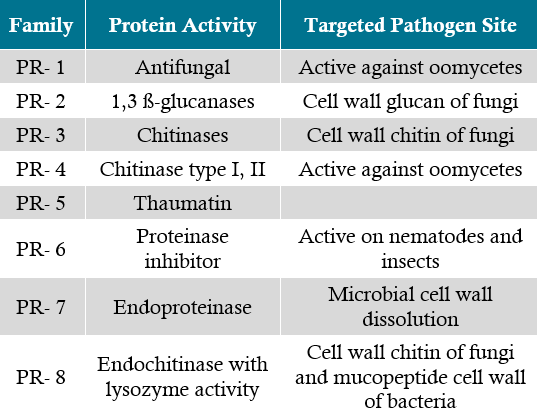
Some pathogens, like many necrotrophs, can infect a large range of plants. If a plant species can be infected by a pathogen, then it falls into the host range. If disease occurs during infection, then that is considered a compatible response (Figure 1). However, not all species within the host range for a pathogen are susceptible. When a pathogen tries to infect a host, and little to no disease results, it is characterized as an incompatible response. The range of effectiveness of incompatible response, in order from high to low, includes: immunity (no disease symptoms), highly resistant (some minor disease symptoms), and highly susceptible (substantial disease symptoms).
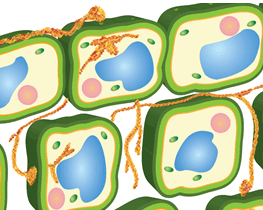
Figure 1. A representation of a plant and a biotrophic pathogen displaying a compatible response.
Plant Detection and Response to Pathogens
Plants have a multifaceted approach for detecting and defending against pathogens. Initial responses are less intensive and use fewer resources. If the situation becomes more severe, then plant defenses can ramp up to counter the threat. Basal resistance, also termed inducible or innate immunity, is the first line of defense against pathogen attack.
When the plant detects the pathogen, it is recognizing microbe-associated molecular patterns (MAMPs). MAMPs can include common components of microbe cell walls, specific proteins, and lipopolysaccharides. Non-pathogens can also trigger basal resistance because their makeup can be detected as a potential threat. However, some pathogens can suppress basal resistance, requiring the plant to resort to more extreme measures of defense.
Hypersensitive response is when the plant deliberately terminates infected cells for the safety of the entire system (Figure 2). It cuts off the pathogen’s access to water and nutrients, removing resources that are necessary for survival and limiting the spread of infection in the plant. This is a more specific response after the recognition of a pathogen effector molecule. Effector molecules selectively bind to proteins and regulate biological activity. They can increase or decrease enzyme activity, gene expression, or cell signaling. In other words, the plant can detect when a foreign molecule is trying to control how a cell is operating.
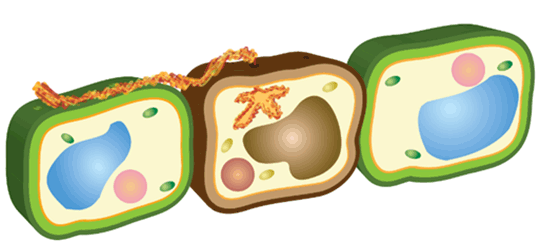
Figure 2. Representation of hypersensitive response in plants, in which infected cells are teminated by the plant to limit the spread of infection.
Once hypersensitive response is triggered and threat of infection is certain, the plant can amplify defenses against a broad range of pathogens for an extended period. This is called systematic acquired resistance (SAR). The ability for the plant to react to new threats is elevated as resources are prepared and mobilized. These resources include chemical compounds, proteins, and enzymes.
For virus protection, some plants can recognize foreign molecules and digest them, so they are no longer a threat. This process is called RNA silencing. In some cases, plants can retain a template, much like an immune system, to discover future threats faster. Soybean plants able to resist soybean mosaic virus and soybean dwarf virus have been developed using transgene-induced RNA silencing. Resistance to these viruses was achieved by transforming plants with genes or gene segments derived from viruses, creating what is referred to as pathogen-derived resistance (Kasai et al., 2013). It has been suggested that methods like this might be useful when looking for resistance in non-viral pathogens as well.
Structural Defenses
The Cell
The cell passively dissuades many potential organisms from establishing, inhabiting, and infecting the plant. Cell walls are thick and can quickly activate chemical defenses at the detection of pathogens (Figure 3). Primary cell walls provide structural support with cellulose. Microfibrils are fibers made of cellulose that allow the cell to be flexible. Hemicellulose is made of cross linking glycans, which provides extra strength to the cell. Pectins act as a glue that keeps neighboring cells together while regulating water content. The combination of these components fortifies the cell. When an element is removed, the stability of the overall structure is compromised, allowing pathogen invasion.
Figure 3. Imbibitional chilling and cold injury. Note club-shaped coleoptile and leafing out underground.
Induced Modifications
Upon detection of pathogens, proteins along the cell wall prepare to protect the cell by inducing defenses. Reactive oxygen species are released to damage as much of the pathogen as possible. A reactive oxygen species, or ROS, has unpaired electrons. This, coupled with the high electronegativity of oxygen, causes these free radicals to take electrons from whatever is closest. Essentially, electrons are pulled from cell walls, degrading the pathogen. The oxidative burst also encourages cross linkage between plant cell wall polymers as electrons are removed. This drastic step in cell defense signals to other components of the plant that there has been a pathogen attack.
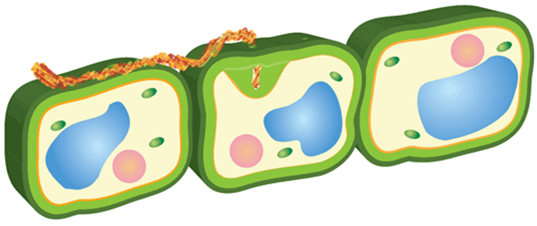
Figure 4. Close-up illustration of the cell wall, showing the key components that maintain structural integrity.
The plant can also synthesize callose in the membrane between the cell wall and the area adjacent to the pathogen. This is called papillae and can either slow down or halt pathogen development (Figure 4). The success of induced modifications depends greatly on the speed at which they are activated.
Tissues and Modifications
Solitary cells can accomplish great feats in plant defense while utilizing individual cell attributes; however, a network of cooperative cells can build larger structures and signal to one another when coordinated action needs to be taken on a greater scale. The goal of modified tissues is to make the plant surface inhospitable for epiphytic pathogen growth. The waxy cuticle of many plant leaves repels water, reducing the potential for moisture to collect and persist on leaves where it can provide a favorable environment for fungal spore germination. Guard cells, which allow gas exchange on plant surfaces, close when in the presence of MAMPs. Trichomes are hairs found on plants that provide a physical and chemical barrier to the plants. Glandular trichomes secrete repellant oils.
Chemical Defense
Chemicals involved in defense are called secondary metabolites, because they are not necessary for the everyday functionality of the plant. These are typically classified as either phytoalexins or phytoanticipins. Phytoalexins are synthesized in response to pathogen attack and phytoanticipins are preformed inhibitors (Freeman and Beattie, 2008). There are many compounds that can be used in either a preformed or induced mechanism for plant defense.
Terpenoids
Terpenoids, or terpenes, are the largest class of secondary metabolites. Different types of terpenoids are defined by how many isoprene units they contain. Isoprene is a volatile gas emitted by some plants during photosynthesis. Plants that produce isoprene are more well-suited to enduring rapid heat stress and are more tolerant to their own reactive oxygen species that they produce (Gould, 2004). A small alteration in the number of isoprene units and their structure can result in a great variation in the defense compounds.
Monoterpenoids are highly volatile compounds that can carry some antibacterial and antifungal activity. Many people are familiar with essential oils and spices, which are comprised of monoterpenoids. Triterpenoids are similar in structures to hormones and sterols. When multiple triterpenoids accumulate, they create saponins, which are incredibly important in plant defense. Saponins are glycosylated triterpenoids, they create a soap-like substance that disrupts the membranes of invading pathogens.
A study conducted on oats demonstrated that mutants deficient in avenacin saponins were susceptible to pathogen invasion via Gaeumannomyces graminis var. tritici (Papadopoulou et al., 1999). This pathogen causes the disease take-all in wheat and barley, but oats are typically resistant to it.
Phenolics are another large class of secondary metabolites involved in plant defense against pathogens. They are found in many different tissues throughout the plant and play a key role in plant defense. Flavonoids are a large subclass of phenolics that can be further divided into subgroups depending on their structure.
Anthocyanins are the compounds responsible for bright colors in plant tissue and provide a multifaceted defense approach. They help the plant protect itself from UV damage by scavenging free radicals, like reactive oxygen (Gould, 2004).
Phytoalexins are isoflavonoids with antibiotic and antifungal properties. These are typically pathogen-specific and disrupt pathogen metabolism or cellular structure.
Tannins are a class of polyphenolic molecules with a diverse range of biochemical functions, including protection against pathogens. Tannins are stored in vacuoles and released when cells are damaged to protect the plant. Tannins are commonly found in roots tissues to protect plants from soil-borne pathogens.
Furanocoumarins are a class of protective compounds common in citrus plants to defend against pathogens and herbivorous insects. Furanocoumarins are activated by UV light and have the potential to be highly toxic. They integrate into the DNA, which can cause rapid cell death.
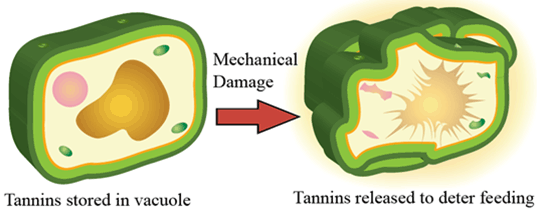
Figure 5. Imbibitional chilling and cold injury. Note club-shaped coleoptile and leafing out underground.
Alkaloids
This class of nitrogen compounds includes some of the most well-known chemicals associated with plant defense such as caffeine and nicotine, which provide protection against pathogens, insects, and herbivores. These substances can cause significant alterations to the physiology of organisms, which can be deadly.
Proteins and Enzymes
Proteins and enzymes are complex structures when compared to chemicals used in plant defense. Therefore, they are only synthesized after pathogen infections. Pathogenesis-related proteins, often called PR proteins, can be acidic or basic and incorporate varying antifungal properties. In addition, PR proteins also aid the plant when it encounters abiotic stresses. There are numerous PR proteins that have been classified, but there is still much to discover (Table 1).
Defensins are a family of PR proteins that can inhibit the growth and functionality of bacteria and fungi by disrupting ion balance. This is accomplished by constraining ion channels or by creating new pores in membranes. Digestive enzyme inhibitors release a protein that inhibits starch digestion. Alpha-amylase inhibitors inhibit protein synthesis, which is incredibly toxic, even to humans. Protease inhibitors interfere with digestion. After herbivory commences, plants can move protease inhibitors to distal areas of the plant, protecting the unharmed areas. Hydrolytic enzymes can degrade the cell walls of pathogens. Chitinases destroy chitin, which composes the cell wall of true fungi. Glucanases degrade glucans, which are what protect oomycetes (water molds). Lysosomes can damage bacterial cell walls.
Table 1. Imbibitional chilling and cold injury. Note club-shaped coleoptile and leafing out underground.
Qualitative vs Quantitative Resistance
An understanding of disease resistance must be coupled with an understanding of genetics. There are two ways in which resistance can be expressed in plant genomes, qualitative and quantitative. Qualitative resistance yields phenotypes that follow basic Mendelian genetics and can be viewed as dominant or recessive. Quantitative resistance, on the other hand, is the product of many traits and aspects of a plant. Disease resistance can be expressed both ways.
For example, wheat displays both quantitative and qualitative resistance against powdery mildew. A study on durum wheat demonstrated that it has 60 genes associated with disease resistance. The researchers found that in environments with low disease pressure, qualitative traits are enough to defend the plant. However, when more disease is present, resistance genes are expressed in a quantitative manner (Marone, 2013). The efficacy of qualitative resistance traits is often short-lived because of the rapid adaptability of pathogens to single traits. An article published by the American Society of Plant Biologists puts it this way, “[in nature] quantitative resistance genes are responding to the blend of pathogens in a specific environment rather than a single predominant pathogen” (Corwin and Kliebenstein, 2017).
The challenge for improvement of crop varieties is determining how quantitative resistance genes apply to a specific pathogen. This has been the subject of extensive investigation, especially for important agronomic crops. The results from these studies are not necessarily straightforward. For example, analysis of corn mechanisms of resistance to Fusarium verticillioides, found that numerous chemical compounds, enzymes, and proteins were utilized. Structural enhancement was accomplished through the lignification of tissues. Defense responses also included the biosynthesis of many secondary metabolites such as shikimate, phenylpropanoid, flavonoid, terpenoid, and diterpenoid (Wang, et al., 2016).
Pathogen Resistance
Although plants can utilize a range of techniques to defend themselves, some pathogens are still able to create a compatible response and cause infection. They accomplish this with many different strategies that specialize in combating plant defense. Some pathogens can either metabolize or detoxify reactive oxygen species produced by the plant. To slow the accumulation of PR proteins, the pathogen will release fewer elicitor proteins. Some pathogens can exclude chitin from the cell walls of their infection apparatus, causing the surveillance system of the plant to misperceive the pathogen.
Pathogens can also release elicitors that compete with the host receptors that bind to and identify chitin, once again concealing the pathogen. Strong, lignified cell walls can be degraded by some pathogens when they produce a higher concentration of pectolytic enzymes. The rapid degradation of pectin decreases structural integrity and leads to failure of plant defense. In a similar way, pathogens can use stronger hydrolytic enzymes, like esterases, to split esters that results in the degradation of plant cells.
In other words, even if the plant is doing everything right, it can still fall victim to infection by a pathogen. As plants evolve more successful defenses, they increase the selection pressure on the pathogens. Therefore, the few pathogens that have the tools necessary to overcome plant defenses persist and multiply. The cycle begins anew as plants develop additional methods to counteract this. These interactions are known as boom and bust cycles (Figure 6).
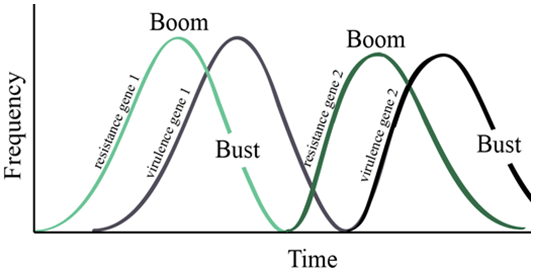
Figure 6. Imbibitional chilling and cold injury. Note club-shaped coleoptile and leafing out underground.
Conclusions
When it comes to defending themselves, plants have many tools at their disposal. Preformed defenses deter many pathogens, insects, and even larger vertebrates from damaging the plant. They can contain highly perceptive surveillance systems that are able to detect pathogens even before infection takes place. Induced defenses can be deployed to shut down the progression of infection before it can affect the whole system. However, pathogens have their own means to survive by preventing, stalling, or overpowering plant defenses. Evolution has armored both plants and pathogens with competing structures, enzymes, and communication systems. When plant breeders are evaluating potential gene pools to incorporate into crop hybrids and varieties, they are assessing a multifaceted approach to overcoming disease. No single mechanism of plant defense can be relied upon exclusively. Instead, a utilization of the plant’s diverse defense capabilities is more likely to achieve success. Breeders work to understand and incorporate these attributes into crop cultivars, making them more resistant to common crop diseases.
References
- Corwin, J.A., and D.J. Kliebenstein. 2017. Quantitative resistance: more than just perception of a pathogen. The Plant Cell 29:655-665.
- Devi, E.L., S. Kumar, T.B. Singh, S.K. Sharma, A. Beemrote, C.P. Devi, S.K. Chongtham, C.H. Singh, R.A. Yumlembam, A. Haribhushan, N. Prakash, and S.H. Wani. 2017. Adaptation strategies and defence mechanisms of plants during environmental stress. Med. Plants. Environ. Chall. pp. 359-413.
- Freeman, B.C. and G.A. Beattie. 2008. An Overview of Plant Defenses against Pathogens and Herbivores. Plant Pathology and Microbiology Publications. 94.
- Gould, K.S. 2004. Nature's Swiss Army Knife: The Diverse Protective Roles of Anthocyanins in Leaves. J. Biomed. Biotechnol. 5:314-320.
- Kasai, M., M. Tsuchiya, and A. Kanazawa. 2013. Gene Duplication and RNA Silencing in Soybean. In J.E. Board (ed) A Comprehensive Survey of International Soybean Research - Genetics, Physiology, Agronomy and Nitrogen Relationships. pp 507-530.
- Marone, D., M.A. Russo, G. Laidò, P. De Vita, R. Papa, A. Blanco, A. Gadaleta, D. Rubiales, and A.M. Mastrangelo. 2013. Genetic basis of qualitative and quantitative resistance to powdery mildew in wheat: from consensus regions to candidate genes. BMC Genomics. 14:562.
- Moosa, A., A. Farzand, S.T. Sahi, and S.A. Khan. 2017. Transgenic expression of antifungal pathogenesis-related proteins against phytopathogenic fungi – 15 years of success, Israel J. of Plant Sci. DOI: 10.1080/07929978.2017.1288407.
- Oerke, E.C. 2005. Crop losses to pests. J. of Agric. Sci. 144:31-43
- Papadopoulou, K., R.E. Melton, M. Leggett, and M.J. Daniels. 1999. Compromised disease resistance in saponin-deficient plants. Proc. Natl. Acad. Sci. USA 96:12923-12928.
The foregoing is provided for informational use only. Contact your Pioneer sales professional for information and suggestions specific to your operation. Product performance is variable and subject to any number of environmental, disease, and pest pressures. Individual results may vary. Pioneer® brand products are provided subject to the terms and conditions of purchase which are part of the labeling and purchase documents.