Country Selector
Please enter a valid location
Propylene Glycol in Silage and Ketosis Prevention
Shared from Inside the ZONE® newsletter, Pioneer Nutritional Sciences
Key Points
- Use of propylene glycol (or 1,2 propanediol, PG) in silage as prevention of metabolic disease in early lactation dairy cows.
- Even though this is not new information, there is some noise in Europe about this topic. Marketing folks are using PG as a “plus”, a differentiator to use in their sales pitch. We at Pioneer have looked into this with our inoculants. However, we don’t want to give false hopes to our customers, since it is not possible to guarantee high enough PG levels to be effective.
- Intro
- Background
- When Does Trouble Occur?
- What’s the Role of PG?
- What About the Silage Content of PG?
- Can PG in Silage Prevent Ketosis?
- Can PG Prevent or Treat Ketosis?
- References
Intro
You will probably see the following claims in the field soon, as university researchers have been approached by companies looking to bang this drum in the US. Captures from their web sites include:
Schaumann GMBH, represented in US by Provita Supplements:
“The silage inoculant BONSILAGE FIT reduces the risk for secondary fermentation by shifting the fermentation acids to more acetic acid and less lactic acid at similar forming of propylene glycol. This process supports the fitness of the cows and reduces the risk for acidosis and ketosis. This has a very positive effect on the energy metabolism of the animals and improves insemination index and calving interval.”
Lallemand, claims in Asia Pacific market for Lalsil include:
“Higher energy value thanks to the metabolic pathway of Lactobacillus buchneri NCIMB 40788 results in improved animal performance.”
Background
Following calving, most high producing cows will go through a period of negative energy balance (NEB). This is inevitable because the nutrient and energy demand for milk production increases rapidly and can’t be fulfilled by intake alone. It is estimated that glucose demand increases from 1 kg/d in late gestation, to 2.5 kg/d right after calving. The cow adapts by mobilizing reserve tissues to meet this demand. Stored fat is released into circulation as non-esterified fatty acids (NEFA), then picked up by the mammary gland for synthesis of milk fat or by the liver where it is oxidized to obtain energy.
When Does Trouble Occur?
Excessive fat mobilization can overwhelm the liver, leading to disease. The incomplete oxidation of NEFA releases ketone bodies into the blood stream (ketosis) and accumulation of triacylglycerol in the liver (fatty liver). Disease around the onset of lactation has lasting negative effects in cow performance. Reports indicate milk loss of more than 500 kg over the lactation, and poor reproductive success.
What’s the Role of PG?
Excessive fat mobilization can overwhelm the liver, leading to disease. The incomplete oxidation of NEFA releases ketone bodies into the blood stream (ketosis) and accumulation of triacylglycerol in the liver (fatty liver). Disease around the onset of lactation has lasting negative effects in cow performance. Reports indicate milk loss of more than 500 kg over the lactation, and poor reproductive success.
What About the Silage Content of PG?
Lactic acid bacteria (LAB) in silage inoculant convert sugars present in the forage into lactic acid thus rapidly reducing the pH. Under this condition, plant enzymes become inactivated and spoilage microorganisms like yeasts, clostridia and molds are inhibited.
Lactobacillus buchneri in absence of oxygen, degrade moderate amounts of lactic acid to acetic acid, 1,2 propanediol (PG), and traces of ethanol1. This is the origin of the competitor’s claim.
According to Drs. Limin Kung and Rich Muck2, PG can be found in silages inoculated with L. buchneri. However, moderate accumulation only happens after 30 to 60 days into fermentation because the metabolism of lactic acid is not fully activated before this time. Normally, the range of PG is 0.25 to 1.5% and concentrations as high as 4.9% have been observed after 120 days fermentation in small scale lab conditions3.
In addition, other species of Lactobacillus (L. diolivorans, and L. reuteri), normally found in silage, can degrade PG4,5, further reducing the possibility any accumulation of PG at a biologically significant level in the silage.
In summary, there is no way to know for sure the level of PG in the silage at any point, because synthesis and degradation occur at the same time.
We protect our customers by not promising what we can’t guarantee.
Can PG in Silage Prevent Ketosis?
There is no doubt that PG in silage contributes to the cow’s pool of propionate for glucose synthesis. However, let’s do a quick calculation:a cow eating 25 kg DM (~55 lb) of TMR that is 50% silage (12.5 kg or 28 lb) that has 1% PG would consume 125 grams of PG per day. Assuming there is no PG degradation in the bunk. This is just a fraction of the recommended bolus dose for ketosis treatment (250 to 400 grams)6. Moreover, since the first sign of ketosis in a cow is usually being off feed, then PG consumption from silage would be even less.
Can PG Prevent or Treat Ketosis?
In 2013, the economic loss due to subclinical ketosis (SCK) was estimated to be between $46 and $92 per case7. Clinical ketosis is defined as BHBA >3 mM/L, cow signs are depressed appetite, weight loss, and decreased milk production, subclinical ketosis is when BHBA is 1.2 to 2.9 mM/L. There is abundant evidence in the literature of PG effectiveness on treating ketosis, reducing NEFA and BHBA in plasma as a bolus dose (drench)8. Contrary to Europe, in the US oral drenches are rarely used in a routine basis, mainly because of the practical aspects and the labor involved. Research aimed to simplify its use by incorporation of PG in the TMR (top dressed at doses of: 2.5 mL per kg of metabolic body weight9, 400 mL/d10, or 600 mL/d11) to date have not shown any effects. Researchers speculate PG absorption is related to the rate its consumed11. When fed, not enough PG is absorbed to have any metabolic effects.
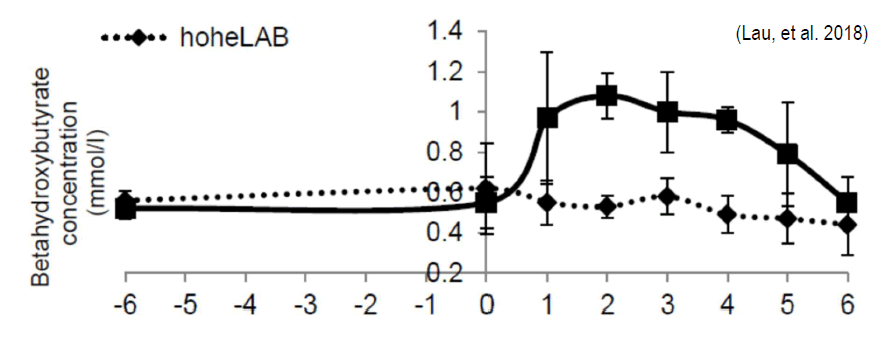
The piece of research that Schaumann’s claim is based upon is below12. Keep in mind that to date this experiment has not been published in a peer reviewed journal. However, it was presented at the Silage Conference held in Bonn, Germany in 2018. Both 1st and 2nd authors are listed as Shaumann employees. Grass silage was inoculated with 2 mixtures of LAB, fermented for 90 d and fed to 2 groups of 60 cows (12.000 kg/lactation) for 3 months. Blood samples were taken 6 weeks before calving, at calving and weekly. The content of PG in control silage was 0.06% (0.4% TMR) and treated silage 3.9% (1.7% TMR).
The chart shows BHBA measured in plasma. There was an effect of treatment (p<0.001), over the 6 weeks control cows BHBA averaged 0.25± 0.09 mM/L, while cows fed treated silage averaged 0.61± 0.18 mM/L. Note that no cows were above 1.2 mM/L (the threshold for sub clinical ketosis). The chart reports data of only 11 cows per treatment, however there were 120 total enrolled cows, with 60 cows per treatment.
References
- Oude Elferink, S.J., J. Krooneman, J.C. Gottschal, S.F. Spoelstra, F. Faber, and F. Driehuis. 2001. Anaerobic conversion of lactic acid to acetic acid and 1, 2-propanediol by Lactobacillus buchneri. Appl Environ Microbiol. 67(1):125-32.
- Kung, J., L. and M.R. E. 2018. Silage Additives Don’t Solve Ketosis. Hoard’s Dairyman.
- Nishino, N., M. Yoshida, H. Shiota, and E. Sakaguchi. 2003. Accumulation of 1,2-propanediol and enhancement of aerobic stability in whole crop maize silage inoculated with Lactobacillus buchneri. Journal of Applied Microbiology. 94(5):800-807.
- Krooneman, J., F. Faber, A.C. Alderkamp, S. Elferink, F. Driehuis, I. Cleenwerck, J. Swings, J.C. Gottschal, and M. Vancanneyt. 2002. Lactobacillus diolivorans sp. nov., a 1,2-propanedioldegrading bacterium isolated from aerobically stable maize silage. Int J Syst Evol Microbiol. 52(Pt 2):639-646.
- Li, Y. and N. Nishino. 2011. Monitoring the bacterial community of maize silage stored in a bunker silo inoculated with Enterococcus faecium, Lactobacillus plantarum and Lactobacillus buchneri. Journal of Applied Microbiology. 110(6):1561-1570.
- Kung, L., Jr., R.D. Shaver, R.J. Grant, and R.J. Schmidt. 2018. Silage review: Interpretation of chemical, microbial, and organoleptic components of silages. J Dairy Sci. 101(5):4020-4033.
- Oetzel, G. 2013. Understanding the impact of subclinical ketosis. University of Wisconsin: Madison.15-26.
- Overton, T.R. and M.R. Waldron. 2004. Nutritional Management of Transition Dairy Cows: Strategies to Optimize Metabolic Health. Journal of Dairy Science. 87:E105-E119.
- Christensen, J.O., R.R. Grummer, F.E. Rasmussen, and S.J. Bertics. 1997. Effect of method of delivery of propylene glycol on plasma metabolites of feed-restricted cattle. J Dairy Sci. 80(3):563-8.
- Mikuła, R., E. Pruszyńska-Oszmałek, M. Ignatowicz-Stefaniak, P.A. Kołodziejski, P. Maćkowiak, and W. Nowak. 2020. The effect of propylene glycol delivery method on blood metabolites in dairy cows. Acta Veterinaria Brno. 89(1):19-29.
- Chibisa, G.E., G.N. Gozho, A.G. Van Kessel, A.A. Olkowski, and T. Mutsvangwa. 2008. Effects of peripartum propylene glycol supplementation on nitrogen metabolism, body composition, and gene expression for the major protein degradation pathways in skeletal muscle in dairy cows. J Dairy Sci. 91(9):3512-27.
- Lau, N., E. Kramer, and J. Hummel, Impact of grass silage with high levels of propylene glycol on ketosis prophylaxis during transition phase and early lactation, in XVIII International Silage Conference, K. Gerlach and K.H. Südekum, Editors. 2018: Bonn.
Pioneer® brand products are provided subject to the terms and conditions of purchase which are part of the labeling and purchase documents.
