Country Selector
Please enter a valid location
White Mold Management in Soybeans

Crop Insights
Written by Mark Jeschke, Ph.D., Pioneer Agronomy Manager
Key Points
- White mold (Sclerotinia sclerotiorum) is a fungal disease of soybean that has become a more frequent issue over the past 30 years in the Northern U.S. and Canada.
- White mold is a disease of high yield potential soybeans — the better the establishment and growth of the crop, the greater the risk of white mold.
- White mold is favored by cool and wet weather and dense soybean canopies that help retain these conditions under the crop canopy.
- Integrating several cultural practices is the most effective means of managing white mold. Cultural practices include variety selection, crop rotation, weed management, no-till and if necessary, limiting dense canopy formation.
- Several fungicides are labeled for white mold but must be applied before the appearance of symptoms and generally will not provide complete control.
- Foliar chemical applications should be targeted at early flowering (R1); penetration of spray to the lower soybean canopy is necessary for treatments to be effective.
- A Growing Problem in Soybeans
- Life Cycle and Symptoms
- Favorable Conditions
- Management of White Mold
- Video: Soybean Growth Staging for Fungicide Timing
- Fungicide Treatments for White Mold
- Corn and Soybean Scouting Calendar
- References
A Growing Problem in Soybeans
White mold (Sclerotinia sclerotiorum) is a fungal disease that can attack hundreds of plant species. Also known as Sclerotinia stem rot, white mold was first observed on soybeans in central Illinois in 1948 and for many years was only a sporadic soybean disease in Minnesota, Wisconsin and Michigan. However, since the 1990s it has become a more frequent threat to northern states from Minnesota to New York, as well as the northern areas of states bordering to the south.
The reason for the increase in the frequency and severity of white mold infection is not fully understood. Changes in soybean management practices likely have played a role. Practices such as earlier planting, longer maturity varieties and narrow row spacing that have been important in driving higher soybean yields also tend to create a more favorable environment for white mold disease development by accelerating canopy closure during the season. Changes in genetic resistance of commercial soybean varieties, as well as changes in the pathogen itself may also be factors.
A successful management plan for white mold in soybean needs to take factors such as variety selection and agronomic management into account, in addition to any chemical control treatments.

Figure 1. White fungal mycelia visible on the stem of a soybean plant infected with white mold.
Life Cycle and Symptoms
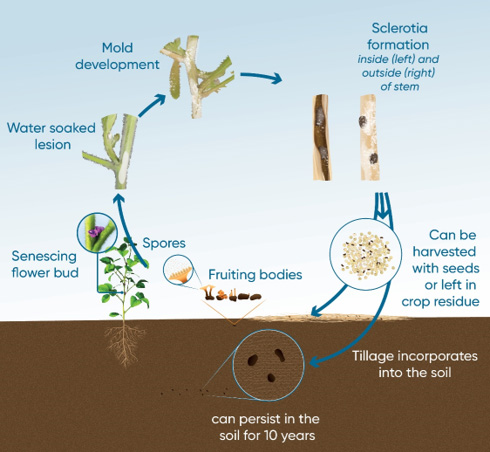
White mold is a monocyclic disease, which means that it goes through one development cycle per crop cycle. White mold persists in soybean fields over time by survival structures called sclerotia. These dark, irregularly shaped bodies about ¼ to ½ inch long are formed within the white, cottony growth both inside and outside the stem. Sclerotia contain energy reserves and function much like seeds, surviving for years in the soil and eventually germinating, producing millions of spores beneath the plant canopy.
In the most common form of germination, a sclerotium produces one or more germ tubes or stipes that grow upward from a depth of two inches or less in the soil. When it reaches the soil surface, the germ tube is triggered by light to produce a small, flesh-colored structure much like a mushroom, called an apothecium. One sclerotium can produce numerous apothecia simultaneously or sequentially throughout the growing season. Each apothecium produces millions of spores beneath the plant canopy, which are periodically released and spread to the plants.
White mold spores are not able to invade plants directly but must colonize dead plant tissue before moving into the plant. Senescing flowers provide a ready source of dead tissue for colonization (Figure 2). Flowers start senescing as soon as they open. From these senescing flowers in the branch axils or stuck to developing pods, the fungus spreads to healthy tissue.

Figure 2. Senescing flowers are the entry point for the white mold pathogen to infect the plant.
It takes around 2 to 3 weeks from initial infection for the fungus to colonize the plant and erupt. The first symptom of white mold infection appears as a water-soaked stem lesion originating from a node. If the lesion remains wet, it will become overgrown with white mold. The disease can then spread directly from plant to plant by contact with this moldy tissue. Sclerotia are formed within the moldy growth and inside the stem to complete the disease cycle (Figure 3).

Figure 3. White mold sclerotia on soybean stem.
The shape of the sclerotia can vary based on where they form. Those that form outside the plant will be more spherical, while those that form inside the plant stem will be more oblong. Plant damage is incurred as tissue rot and formation of sclerotia inside the stem result in rapid wilting and death of the upper part of the plant. As the disease progresses, premature death of the entire plant can occur.
Figure 4. White mold disease cycle. Click here or on the image above for a larger view.
Favorable Conditions
Wet, cool conditions are required throughout the white mold disease cycle, including germination of the sclerotia in the soil, spore release, infection of soybean flowers by spores, and spread of white mold from plant to plant.
- Sclerotia in the soil require 7 to 14 days of high soil moisture to germinate and produce apothecia (fruiting bodies). Temperatures between 40 and 60°F are optimal for this process.
- Spores are forcibly ejected from the fruiting bodies during wet weather conditions.
- After spores are released, a wet surface on senescing flowers or other dead or dying tissue is required for spore germination. Specifically, 2 to 3 days of continuous wetness, or more than 12 hours of daily wetness for 3 to 5 days is required.
- White mycelial growth develops on stem lesions that remain wet, and spreads by contact to neighboring plants. Temperatures under 85°F are favorable for disease spread.
| Wet, cool conditions are required throughout the white mold disease cycle. |
Early establishment of a dense soybean canopy increases the likelihood that the high-humidity conditions required for white mold development will occur. Early canopy closure is a goal for many soybean producers, especially in northern locations and growing environments where solar radiation may be limited, as it important for maximizing light interception and yield. Soybean management practices such as early planting and narrow rows can help achieve earlier canopy closure. Unfortunately, these practices can also encourage white mold development.

Figure 5. White mold sclerotia on soybean stem.
Risk Factors for White Mold
The North Central Plant Health Initiative has developed the following list of risk factors for white mold.
Seasonal Risk Factors for White Mold Development
Weather: Moderate temperatures (<85°F), normal or above normal precipitation, soil moisture at field capacity or above and prolonged morning fog and leaf wetness (high canopy humidity) at and following flowering into early pod development.
Early canopy closure due to early planting, high plant population, narrow rows, excessive plant nutrition and optimal climatic conditions creates dense canopy and increased apothecia density.
History of white mold in the field, density of the white mold pathogen, apothecia present on soil surface at flowering, distribution of pathogen/disease in field.
Soybean variety planted. Plant structure and physiological functions govern variety reaction to white mold. Varieties range from partially resistant to highly susceptible.
Long-Term Risk Factors for White Mold Development
Field/cropping history. Pathogen level will gradually increase if:
- Other host crops are grown in rotation with soybean.
- 1- to 2-year intervals occur between soybean crops.
- White mold susceptible varieties are grown.
Weed management systems. Inoculum will increase if control of broadleaf weeds is ineffective. Some herbicides used in rotation systems may be suppressive to white mold.
Topography of field. Pockets of poor drainage, tree lines and other natural barriers that impede air movement will create a favorable microenvironment for white mold development.
Pathogen introduction:
- Contaminated and infected seed.
- Movement of infested soil with equipment.
- Wind-borne spores from apothecia from area outside fields.
Management of White Mold

Figure 6. Infected soybean stem.
White mold is a disease of high-yield-potential soybeans. Often, the better the establishment and growth of the crop, the more likely it will be damaged by white mold. Management practices that may be useful for reducing the severity of white mold infection may also limit the yield potential of the crop; consequently, an integrated management strategy for white mold often involves weighing the tradeoffs between pushing for maximum yield vs. protecting against disease based on the white mold risk in a given field.
No single practice will be effective in completely controlling white mold, but several options are available to help reduce disease pressure. Current options include disease avoidance, variety selection, changes in cropping systems including tillage and rotation, and adjusting production methods such as planting practices, chemical applications and weed control.
| No single practice will be effective in completely controlling white mold. |
Disease Avoidance
White mold spreads either by movement of spores or sclerotia from field to field. Spores are airborne and may originate from any field that has had white mold in the past. However, spores generally do not move long distances, as they originate near the soil surface and commonly stay contained below the crop canopy. Spread over longer distances is usually due to movement of sclerotia.
Sclerotia move from field to field in harvest equipment or in contaminated seed. Harvest equipment should be thoroughly cleaned when moving from infected to non-infected fields. Harvesting infected fields last provides additional safety. Because sclerotia are roughly the size of soybean seed, they can’t be easily separated by the combine. Soybeans harvested from infected fields are likely to be loaded with sclerotia. Planting these soybeans would place them at the ideal depth for germination and infection of that crop and field. Growers should absolutely not save seed from infected fields.
| White mold spores do not move long distances. Spread from field to field is usually due to movement of sclerotia. |
Corteva Agriscience avoids growing seed beans in fields with a history of white mold. In addition, seed is thoroughly cleaned and inspected to ensure that it is disease-free. Seed cleaning with a gravity table or centrifugal tower is essential to remove sclerotia. Fungicide seed treatments can help ensure that no disease is transmitted by mycelia present on seed.
Variety Selection
There is no absolute resistance available to white mold (all varieties can get the disease under severe pressure), but differences in tolerance exist between varieties. Pioneer variety ratings range from 2 to 7 on a scale of 1 to 9 (9 = resistant). Ratings reflect varietal differences in the rate at which infection develops as well as the extent of damage it causes and are based on data from multiple locations and years. Choosing varieties that rate high for tolerance is an important management practice in areas that commonly encounter white mold. Your local Pioneer sales professional can suggest white mold tolerant varieties with a complete package of traits needed for top soybean production in your area.
Variety maturity is also an important consideration. Longer maturity varieties can help maximize yield potential, but they also have a longer window of flowering, which extends the period of time that senescing flowers are present and susceptible to infection.
No-Till
Research studies have shown that no-till is generally superior to other tillage systems in limiting white mold development by leaving sclerotia to deteriorate on the soil surface. Sclerotia germinate from the top two inches of soil. Below that depth, they can remain dormant for five or more years. Because of its longevity in the soil, it is difficult to devise a strategy to control white mold with tillage. Deep tillage buries sclerotia from the soil surface but may also bring prior sclerotia into their zone of germination.
Crop Rotation
Rotation with a non-host crop can help reduce disease pressure in a field. Non-host crops include corn, sorghum and small grains. Susceptible crops to avoid in a rotation include alfalfa, clover, sunflower, canola, edible beans, potato and others. Depending on soybean tolerance, field history and other factors, more than one year away from soybeans may be required. Including a small grain crop in the rotation can be particularly helpful, as the canopy is dense enough to trigger formation of apothecia from the sclerotia in the soil but there is no host crop to infect. However, because of the longevity of sclerotia in the soil, crop rotation is only a partial solution.
Planting Date
Later planted soybeans are generally shorter and less branched and therefore later to reach canopy closure. Some planting date studies show that later planting results in less incidence of white mold. However, yields are generally reduced when planting is delayed past mid-May in northern states. The tradeoff between less yield reduction due to white mold but more yield reduction due to late planting may not be favorable, especially in years of low disease pressure.
Row Spacing and Seeding Rate
Row spacing and seeding rate both influence soybean canopy closure and density, which affect development of white mold. However, given that early canopy closure is generally favorable to yield, adopting wider row spacings or lower seeding rates to manage white mold may also reduce yield potential.
The most common row spacings for soybeans in the U.S. are 15 inches and 30 inches. Drilled soybeans in row spacings less than 15 inches were once common but have declined in recent years (Jeschke, 2026). Numerous studies over many years have demonstrated a yield advantage for narrow-row (<30 inches) soybeans.
Research has shown that seeding rate is likely a more important factor affecting white mold development than row spacing (Lee et al., 2005). In fields with high risk of white mold, seeding rates should be sufficient for uniform stand establishment, but shouldn’t be aggressively high. Actual rates will vary depending on planting date, seedbed conditions, and seed quality. A multi-state university study found that wider rows and reduced seeding rates were both effective at reducing white mold severity, but also reduced soybean yield when white mold did not develop (Webster et al., 2022). Results suggested that wider rows and reduced seeding rates as tactics to manage white mold should be reserved for fields with a history of white mold where disease is likely to occur.
Weed Control
White mold has over 400 plant hosts, including many broadleaf weeds. Host weeds that are also common weed species throughout soybean growing areas include lambsquarters, ragweed, pigweed and velvetleaf. In addition to acting as host to the disease, weeds can also increase canopy density, which favors disease development.
Video: Soybean Growth Staging for Fungicide Timing
Fungicide Treatments for White Mold
Despite the best use of cultural practices to limit the incidence of white mold, weather and other conditions conducive to disease development may still cause heavy infestations. In cases of high disease risk, a foliar fungicide application or a soil application of a biological product may help reduce disease severity and protect soybean yield.
Products labeled for white mold control or suppression include several foliar fungicides (Table 1), a biological fungicide (Contans® fungicide), and the herbicide lactofen (active ingredient in Cobra® herbicide and Phoenix® herbicide).
Table 1. Fungicides labeled for control of white mold in soybeans with an efficacy of “fair” or better (Wise, 2026).
| Fungicide Trade Name | Active Ingredient | White Mold Efficacy |
|---|---|---|
| Aproach® 2.08 SC | picoxystrobin (11) | good |
| Proline® 490 SC | prothioconazole (3) | fair |
| Domark® 230 ME | tetraconazole (3) | fair |
| Topsin-M® | thiophanate-methyl (1) | fair |
| Omega® 500 DF | fluazinam (29) | good |
| Endura® 0.7 DF | boscalid (7) | very good |
| Propulse® 3.34 SC | fluopyram (7), prothioconazole (3) | good |
| Delaro® 325 SC | trifloxystrobin (7), prothioconazole (3) | fair |
| Delaro® Complete 3.83 SC | fluopyram (7), trifloxystrobin (11), prothioconazole (3) | fair |
| Viatude® 2.09 SC | picoxystrobin (11), prothioconazole (3) | fair |
¹Fungicide Resistance Action Committee mode of action groups: Group 1 MBC Thiophanates, Group 3 DMI Triazoles, Group 7 SDHI, Group 11 Qol Strobilurins, Group 29 2,6 Dinitro-anilines.
Chemical treatments generally will not provide complete control of white mold. Reduction of disease in university field trials has ranged from 0 to 60% (Mueller et al., 2015). Consequently, chemical treatments need to be used as part of an integrated management strategy for white mold.
Foliar Fungicides
Optimum application time of fungicides for white mold control in soybeans is the R1 to R2 growth stage, also known as the beginning bloom or first flower stage (Mueller et al., 2015). For much of the U.S. Corn Belt, the R1 stage coincides with the first two weeks of July when the vegetative growth stage is typically about V7 to V10 (Pedersen, 2009). Fungicides applied up to the R3 stage can provide some benefit in reducing white mold.
Fungicides have little activity on established disease and must be applied prior to white mold invasion of senescing flowers. Applications made just prior to pathogen invasion have helped reduce disease severity in some studies. Because soybeans normally flower for 30 days or more (R1 to R5) and fungicides for white mold control have maximum residual activity of about two weeks, a second application may be necessary if conducive environmental conditions persist into mid-summer.
| Fungicides have little activity on established disease and must be applied prior to white mold invasion of senescing flowers. |
One drawback to later (R3) fungicide application is the potential for reduced canopy penetration. Though soybeans grown in 30-inch rows at moderate seeding rates may allow for good penetration of the lower canopy at R1, spray coverage of the lower nodes becomes increasingly difficult with continued vegetative growth. The lower canopy can remain relatively wet or humid, providing the appropriate environment for pathogenicity. Thus, it is essential for spray droplets to reach the lower twothirds of the soybean canopy in order to obtain satisfactory disease control.
Fungicide Research Results
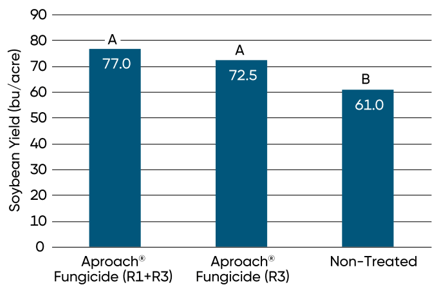
A University of Wisconsin research trial conducted near Hancock, WI in 2016 found significant increases in soybean yield associated with Aproach® fungicide treatment under high levels of white mold pressure (Figure 7). A single treatment at the R3 growth stage increased yield by 11.5 bu/A and sequential applications at the R1 and R3 stages increased yield 16 bu/A compared to the non-treated check.
Figure 7. Yield of soybeans treated with Aproach® fungicide at the R3 growth stage and the R1 and R3 stages compared to non-treated soybeans in a Univ. of Wisconsin trial at Hancock, WI, in 2016 (Smith et al., 2016). Click here or on the image above for a larger view.
Means labeled with the same letter are not significantly different based on Fisher’s Least Significant Difference (LSD; α=0.05).

Figure 8. Soybean plants infected with white mold.
Corteva Agriscience on-farm research trials were conducted in 2017 at locations near Orchard, NE and Edgar, WI that experienced high white mold pressure. Both trials compared sequential applications at the R1 and R3 growth stages and single-pass treatments at both R1 and R3 to a non-treated check. The Wisconsin trial was non-replicated, and the Nebraska trial included two replications. The two-pass fungicide program increased yield by an average of 13.3 bu/A in these trials (Table 2). The R3 and R1 treatments increased yield by an average of 8.7 and 6.7 bu/A.
Table 2. Soybean yield associated with Aproach® fungicide treatments in on-farm trials with heavy white mold pressure in Wisconsin and Nebraska.
| Fungicide Treatment | Edgar WI | Orchard NE | Average | Yield Advantage |
|---|---|---|---|---|
| —————— bu/A —————— | ||||
| Aproach® (R1+R3) | 66.6 | 55.9 | 61.3 | +13.3 |
| Aproach® (R3) | 57.7 | 55.6 | 56.7 | +8.7 |
| Aproach® (R1) | 61.9 | 47.4 | 54.7 | +6.7 |
| Non-Treated | 54.8 | 41.2 | 48.0 | |
Pioneer on-farm trials were conducted in Eastern Canada in 2023 to evaluate the effectiveness of Viatude® fungicide (picoxystrobin + prothioconazole) for the control of white mold in soybeans. The 2023 growing season was very favorable for white mold development in the study area in Eastern Canada. Soybeans treated with Viatude® fungicide yielded an average of 3.2 bu/A more than non-treated soybeans in 30 paired comparisons across 10 locations.
Figure 9. Corteva Agriscience on-farm fungicide research trial near Orchard, NE comparing Aproach® fungicide applied at R1, R3 and R1+R3 growth stages to a non-treated check under heavy white mold pressure (August 23, 2017) Click here or on the image above for a larger view.
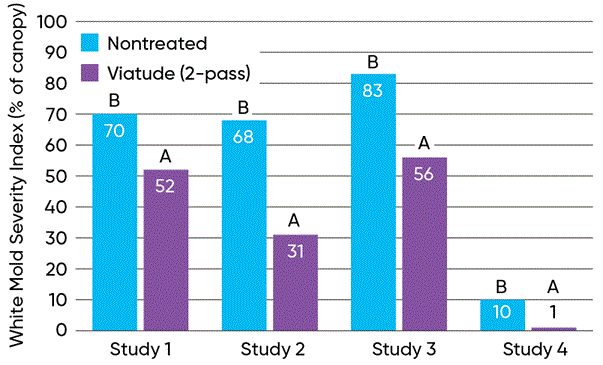
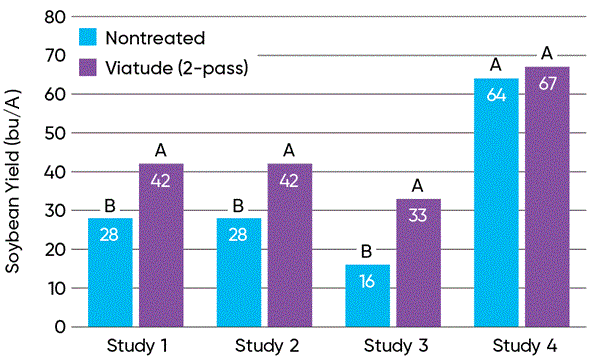
Figure 10. White mold disease severity and yield of soybeans treated with two passes of Viatude® fungicide compared to non-treated soybeans in North Dakota State University trials at Carrington, ND in 2024 (Wunsch et al., 2025). Click here or on the image above for a larger view.
Studies conducted by North Dakota State University at the Carrington Research Extension Center in 2024 evaluated the efficacy of a two-pass foliar fungicide treatment program using Viatude™ fungicide for reducing white mold severity and protecting soybean yield. Four field studies were conducted, three of which experienced very high levels of white mold disease. The two-pass fungicide program significantly reduced white mold severity in all four studies and increased soybean yield in the three studies with high white mold pressure (Figure 10). Fungicide treatment increased yield by as much as 17 bu/A and by an average of 12 bu/A across all four studies.
Biological Control
Contans® WG fungicide is a biological control agent of white mold. The product contains the soil fungus Coniothyrium minitans, which acts as a parasite attacking the overwintering survival structures (sclerotia) of white mold. Contans fungicide is applied to the soil, its spores germinate with sufficient moisture, and the fungus can destroy sclerotia if given adequate time. Contans fungicide should be applied at least three months prior to white mold infection, and soil-incorporated immediately following application to a depth of at least 4 inches. Although Contans fungicide may be fall- or spring-applied, fall applications have performed better than those done in spring.
Corn and Soybean Scouting Calendar
Download your copy of Pioneer's Corn and Soybean Scouting Calendar. It's your guide to the diseases and pests that threaten your yield and when to look for each one.
References
- Agrios, G. N. 1988. How plants defend themselves against pathogens. 97-115. In Plant Pathology, third edition. Academic Press, Inc. San Diego, CA.
- Dann, E. K., B. W. Diers, and R. Hammerschmidt. 1999. Suppression of sclerotinia stem rot of soybean by lactofen herbicide treatment. Phytopathology 89:598-562.
- Jeschke, M. 2026. Row Spacing in Soybean Production. Pioneer Crop Insights. Vol. 36 No. 5. Corteva Agriscience. Johnston, IA.
- Kyle, D. 2014. Effect of Cobra® Herbicide on Soybean Yield in the Absence of White Mold or Weed Pressure. Pioneer Agronomy Research Update. Vol. 4 No. 36.
- Lee. C.D., K.A. Renner, D. Penner, R. Hammerschmidt, and J.D. Kelly. 2005. Glyphosate-resistant soybean management system effect on Sclerotinia stem rot. Weed Technol. 19:580-588.
- Mueller, D., C. Bradley, M. Chilvers, P. Esker, D. Malvick, A. Peltier, A. Sisson, and K. Wise. 2015. White Mold. Soybean Disease Management CPN-1005. Crop Protection Network.
- Nelson, K. A., K. A. Renner, and R. Hammerschmidt. 2001. Effects of protoporphyrinogen oxidase inhibitors on soybean (Glycine max L.) response, Sclerotinia sclerotium disease development, and phytoalexin production by soybean. Weed Technology 16:353-359.
- Nelson, K. A., K. A. Renner, and R. Hammerschmidt. 2002. Cultivar and herbicide selection affects soybean development and the incidence of Sclerotinia Stem Rot. Agron. J. 94:1270-1281.
- Oplinger, E. S., C. R. Grau, J. E. Kurle, J. M. Gaska, and N. Kurtzweil. 1999. Foliar treatments for control of white mold in soybean.
- Pedersen, P. 2009. Soybean growth and development. Iowa State University Extension.
- Smith, D., S. Chapman, and B. Mueller. 2016. Wisconsin Field Crops Pathology Fungicide Tests Summary. Univ. of Wisconsin Extension.
- Webster, R.W., M.G. Roth, B.D. Mueller, D.S. Mueller, M.I. Chilvers, J.F. Willbur, S. Mourtzinis, S.P. Conley, and D.L. Smith. 2022. Integration of row spacing, seeding rates, and fungicide applications for control of sclerotinia stem rot in Glycine max. Plant Disease. 106:1183-1191.
- Wise, K. 2026. Fungicide Efficacy for Control of Soybean Foliar Diseases. Soybean Disease Management CPN-1019-W. Crop Protection Network.
- Wunsch, M., J. Hafner, S. Kallis, A. Fauss, and T. Miorino. 2025. Comparative fungicide efficacy: Management of white mold in soybeans. North Dakota State University Carrington Research Extension Center.
Protect Your Field and Your Yield
Prevent against and defeat even the most resistant diseases with fungicide solutions from Corteva Agriscience.
The foregoing is provided for informational use only. Please contact your Pioneer sales professional for information and suggestions specific to your operation. Product performance is variable and depends on many factors such as moisture and heat stress, soil type, management practices and environmental stress as well as disease and pest pressures. Individual results may vary. Pioneer® brand products are subject to the terms and conditions of purchase which are part of the labeling and purchase documents.